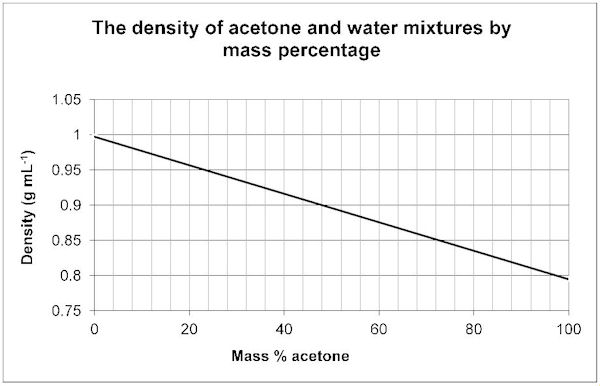
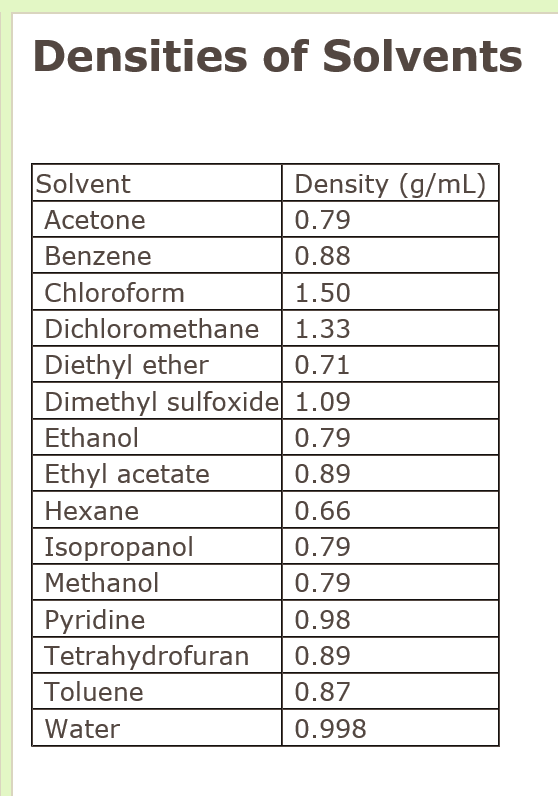
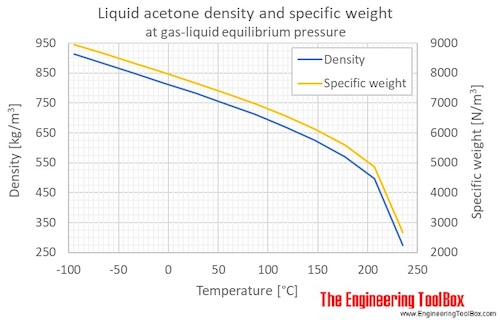
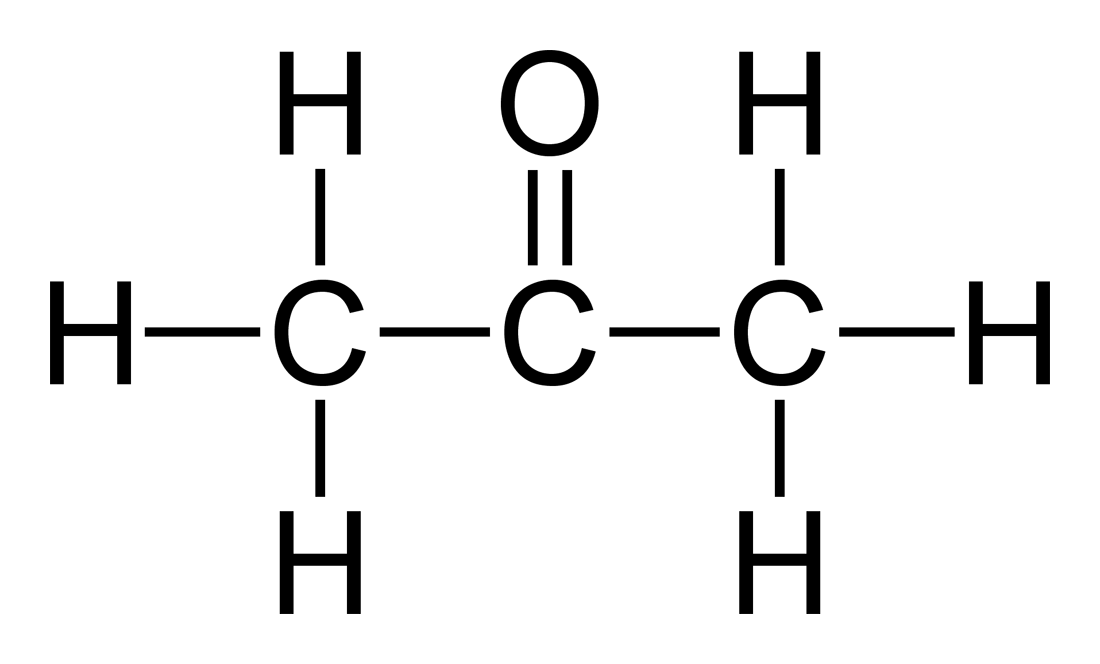
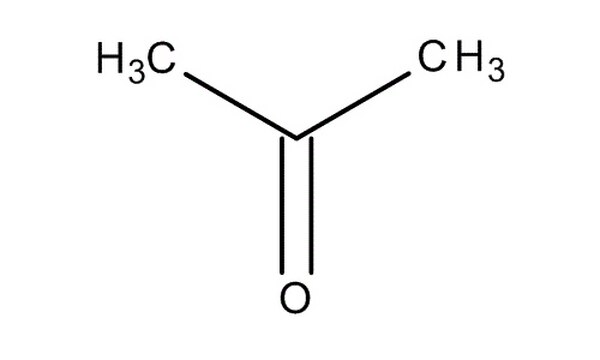
Acetone, the solvent in nail polish remover, has a density of 0791 g/mL What is the volume, in mil - YouTube
Solved] 1. The density of acetone is 0.702 g/wrvrvi'|~_'.~ How much in grams does 9.85 x 10 -2 L mass? (Report answer in 4 SF). 3pts 2. If the speci... | Course Hero

OneClass: The solute is acetone. The density of acetone is .791g/ml. The solvent is acetic acid. The ...

SOLVED:beaker contains 31.5 mL of acetone (C;H;O, density is 0.78 g/mL) Determine how many molecules of acetone are in the beaker: N = : TOOLS x10y