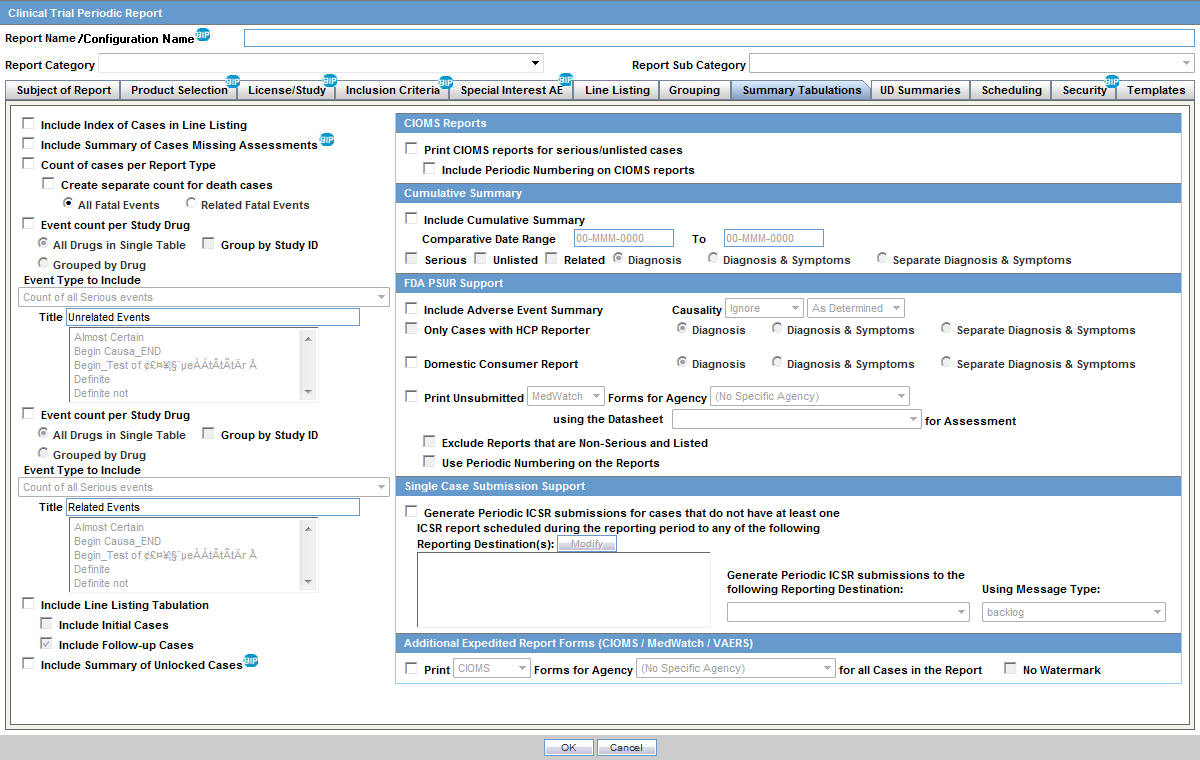
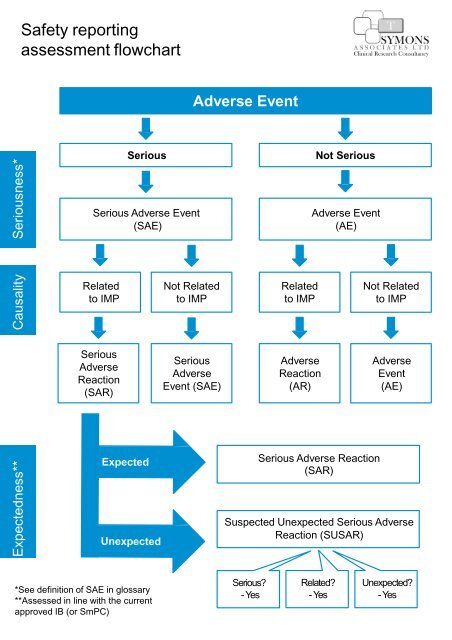
Safety Reporting Overload in Clinical Trials: FDA and Site Perspectives on Overreporting of Adverse Events | CenterWatch

Training log for new clinical research associates (CRAs). P&P, policies... | Download Scientific Diagram

European Medicines Agency on LinkedIn: Clinical Trials Information System: training and support - European…