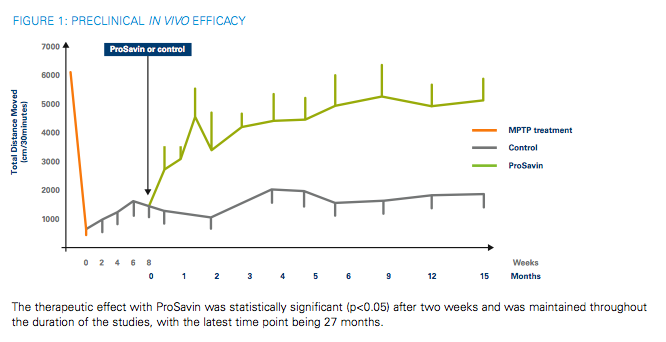
Table 2 from Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: a dose escalation, open-label, phase 1/2 trial | Semantic Scholar
![PDF] Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: a dose escalation, open-label, phase 1/2 trial | Semantic Scholar PDF] Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: a dose escalation, open-label, phase 1/2 trial | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c65a8595b06e893fa131ecf8e90e4bde2d1387b0/2-Figure1-1.png)
PDF] Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: a dose escalation, open-label, phase 1/2 trial | Semantic Scholar
![PDF] Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: a dose escalation, open-label, phase 1/2 trial | Semantic Scholar PDF] Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: a dose escalation, open-label, phase 1/2 trial | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c65a8595b06e893fa131ecf8e90e4bde2d1387b0/5-Table3-1.png)
PDF] Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: a dose escalation, open-label, phase 1/2 trial | Semantic Scholar

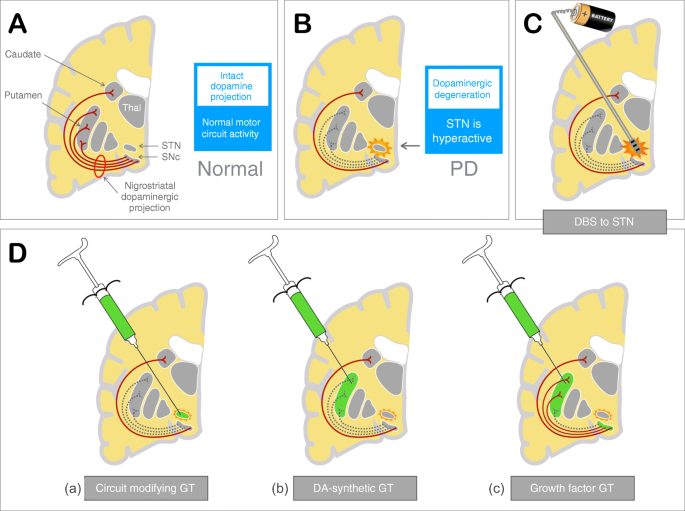
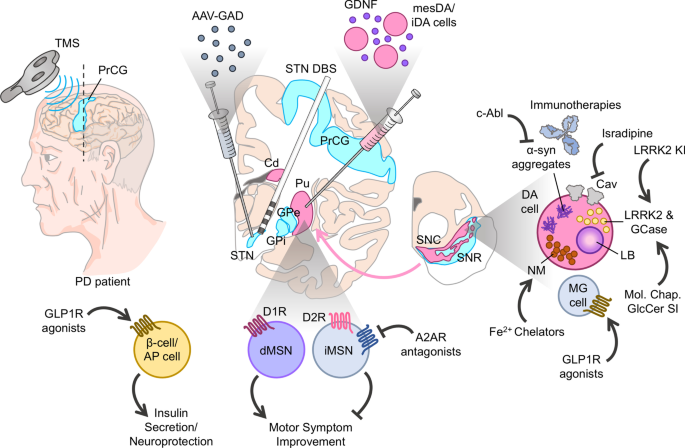
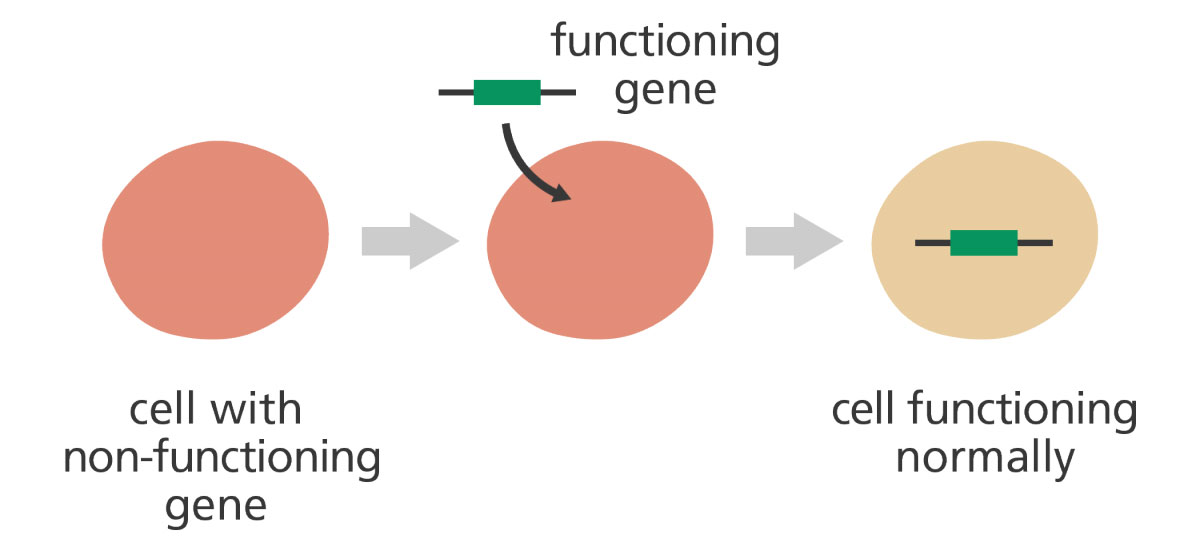
Therapeutic strategies for Parkinson disease: beyond dopaminergic drugs | Nature Reviews Drug Discovery

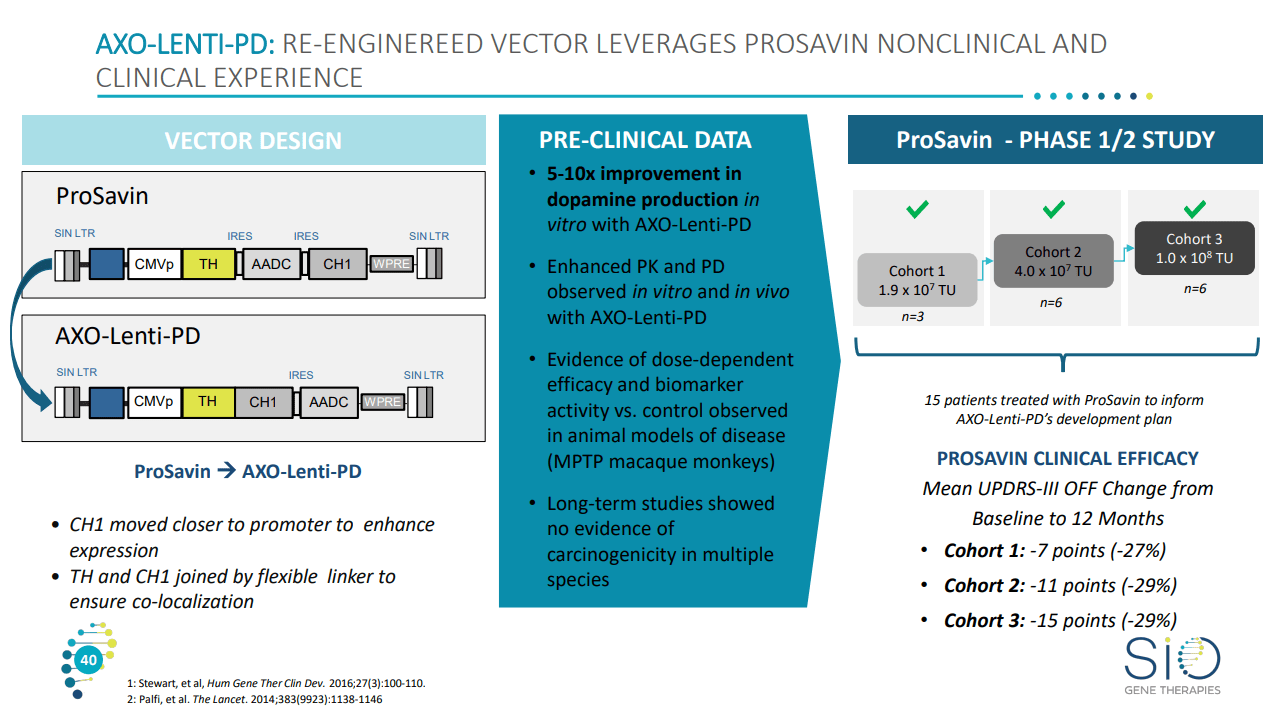
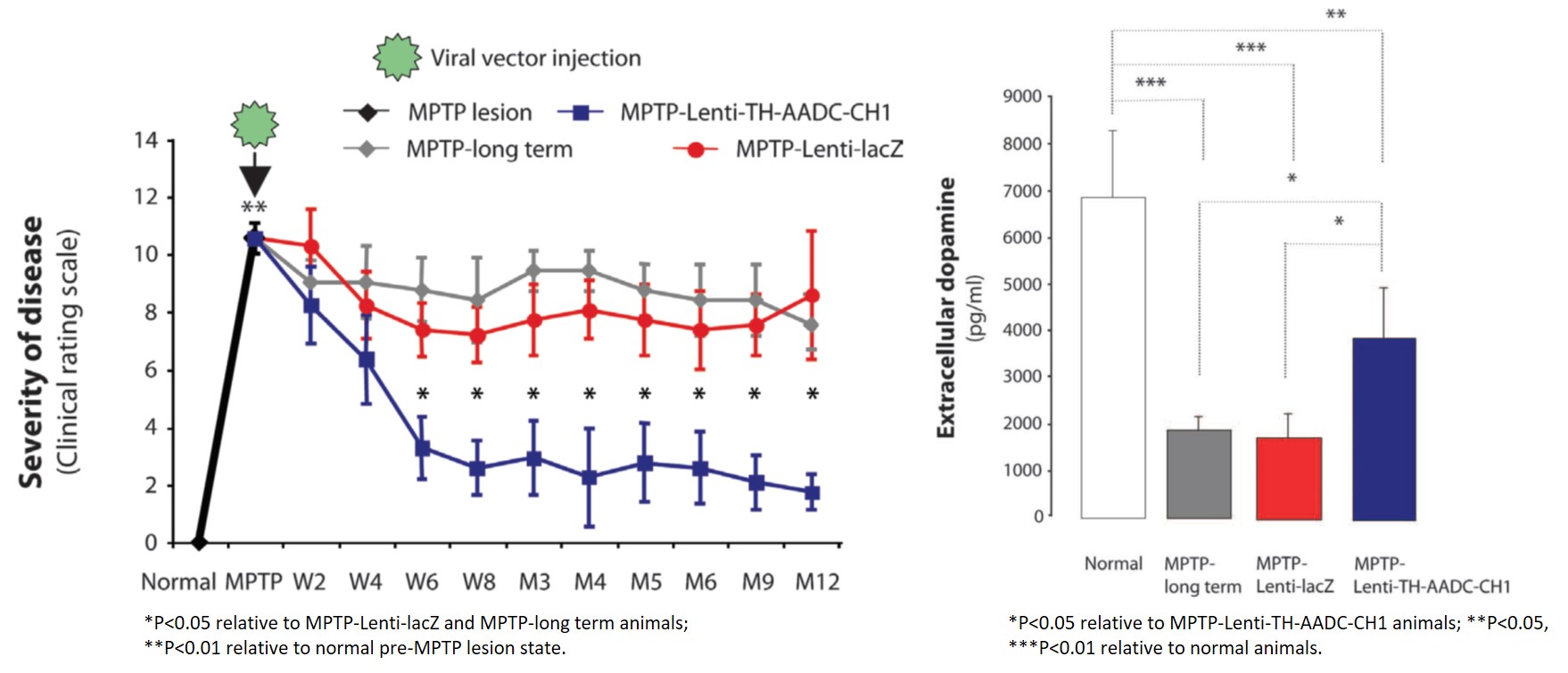
Gene Therapy for Parkinson's Disease: Preclinical Evaluation of Optimally Configured TH:CH1 Fusion for Maximal Dopamine Synthesis - ScienceDirect
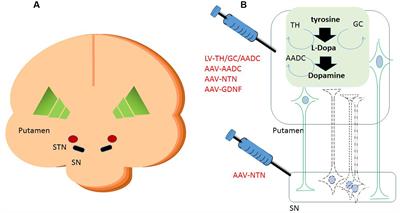
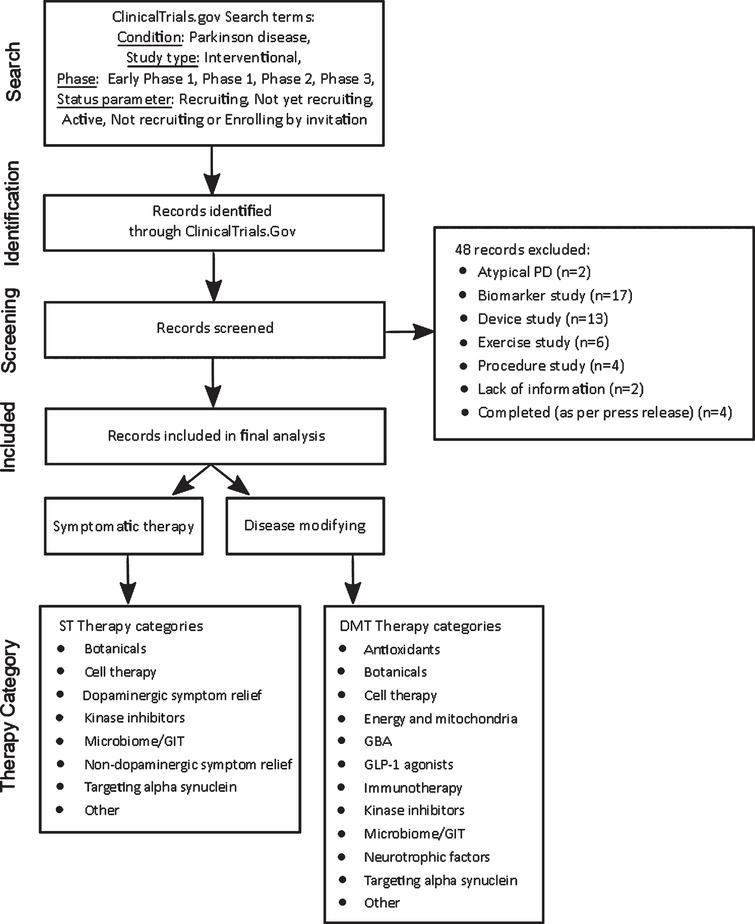
Frontiers | Perspective on the Road toward Gene Therapy for Parkinson's Disease | Frontiers in Neuroanatomy
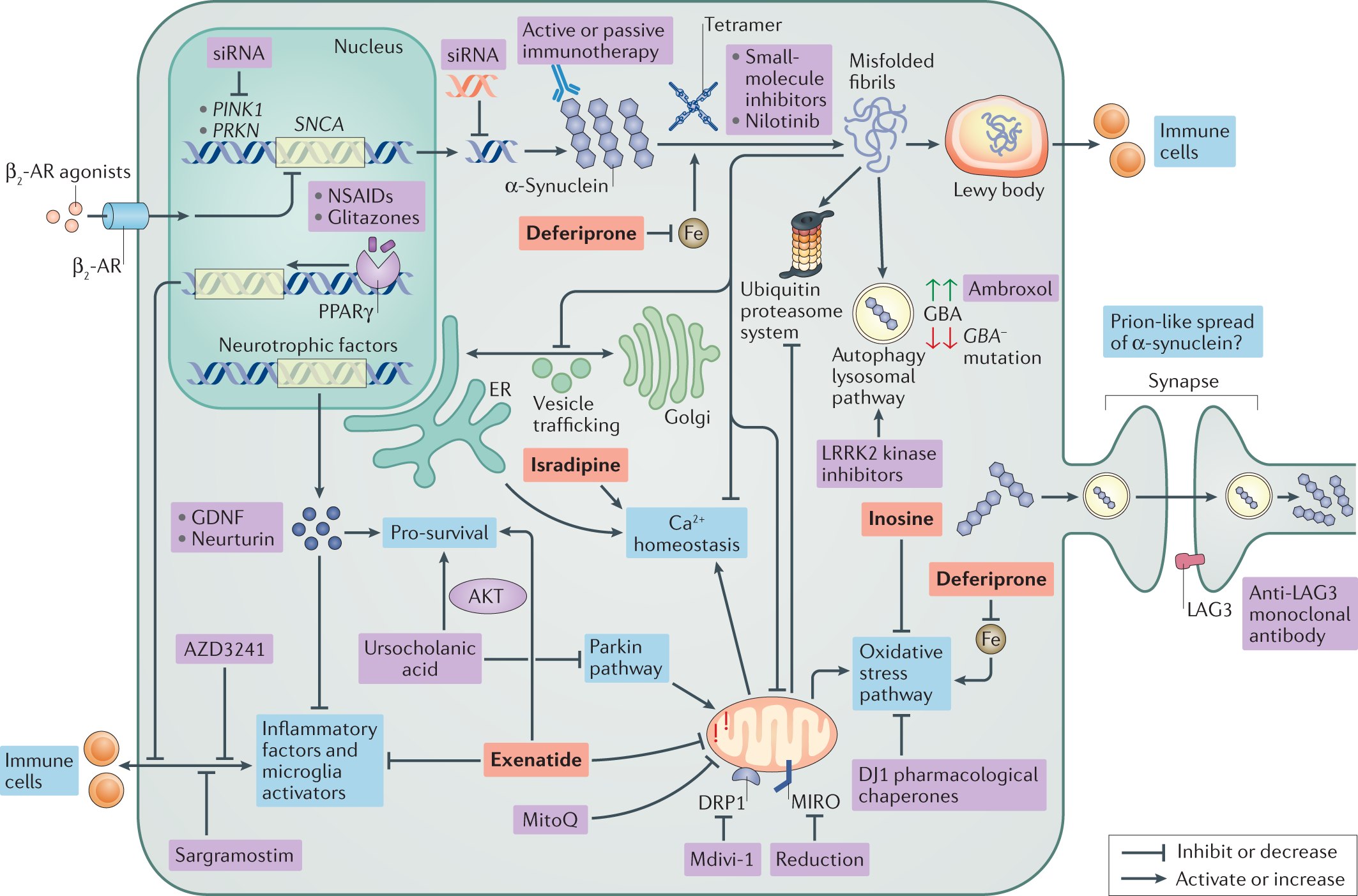
Emerging therapies in Parkinson disease — repurposed drugs and new approaches | Nature Reviews Neurology

Gene Therapy for Parkinson's Disease: Preclinical Evaluation of Optimally Configured TH:CH1 Fusion for Maximal Dopamine Synthesis - ScienceDirect

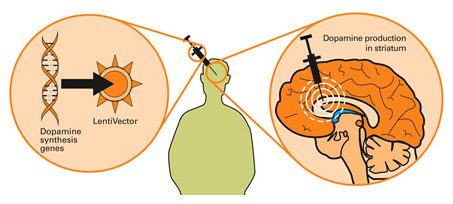
PDF) ProSavin (R) a Gene Therapy Approach for the Treatment of Parkinson's Disease: Phase I Clinical Trial Update

A pie chart of the agents in active phase1 trials for PD, registered on... | Download Scientific Diagram

Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: a dose escalation, open-label, phase 1/2 trial - ScienceDirect

Designing stem-cell-based dopamine cell replacement trials for Parkinson's disease | Nature Medicine
![PDF] Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: a dose escalation, open-label, phase 1/2 trial | Semantic Scholar PDF] Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: a dose escalation, open-label, phase 1/2 trial | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c65a8595b06e893fa131ecf8e90e4bde2d1387b0/3-Table1-1.png)
PDF] Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: a dose escalation, open-label, phase 1/2 trial | Semantic Scholar

ProSavin Trial Results: Once Again, a Gene Therapy Approach to Parkinson's Yields Encouraging Safety Data but Modest Efficacy | Parkinson's Disease

PDF) Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson's disease: A dose escalation, open-label, phase 1/2 trial